Encyclopaedia Britannica, or, a Dictionary of arts, sciences, and miscellaneous literature : enlarged and improved. Illustrated with nearly six hundred engravings > Volume 17, Poetry-RHI
(331) Page 313
Download files
Complete book:
Individual page:
Thumbnail gallery: Grid view | List view
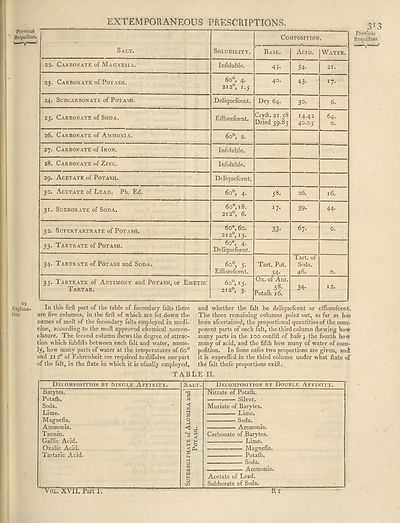
EXTEMPORANEOUS PRESCRIPTIONS.
Previous
Kequifites.
Salt.
22. Carbonate of Magnesia.
23. Carbonate of Potash.
24. Subcarbonate of Potash.
25. Carbonate of Soda.
26. Carbonate of Ammonia.
27. Carbonate of Iron.
28. Carbonate of Zinc.
29. Acetate of Potash.
30. Acetate of Lead. Ph. Ed.
31. Subborate of Soda.
32. Sufertartrate of Potash.
33. Tartrate of Potash.
34. Tartrate of Potash and Soda.
35. Tartrate of Antimony and Potash, or Emetic
Tartar.
Solubility.
Infoluble.
6o°, 4.
212°, I.5
Deliquefcent.
Efflorefcent.
6o°, 2.
Infoluble.
Infoluble.
Deliquefcent.
6o°, 4.
6o®, 18.
2120, 6.
6o°, 60.
212°, 13.
_6o°, 4.
Deliquefcent.
6o°, 5.
Efflorefcent.
6o°, 15.
212°, 3.
Composition.
— A
Base.
45-
40.
Dry 64.
Cryft. 21.58
Dried 59.85
58.
I7*
33-
Tart. Pot.
54-
Ox. of Ant.
38-
Potafh 16.
Acid. Water.
34-
43-
3°-
14.42
40.05
26.
39-
67-
Tart. of
Soda.
46.
34-
21.
I7-
64.
o.
16.
44.
12.
29
Explana¬
tion.
313
Previous
Requifites.
In this firft part of the table of fecondary falls there
are five columns, in the firft of which are fet down the
names of moft of the fecondary falls employed in medi¬
cine, according to the moft approved chemical nomen¬
clature. The fecond column fhewsthe degree of attrac¬
tion which fubfifts between each fait and water, name¬
ly, how many parts of water at the temperatures of 6o°
and 212° of Fahrenheit are required todiffolve one part
of the fait, in the ftate in which it is ufually employed,
TAB
and whether the fait be deliquefcent or efflorefcent.
The three remaining columns point out, as far as has
been afeertained, the proportional quantities of the com¬
ponent parts of each fait, the third column (hewing how
many parts in the 100 confift of bafe $ the fourth how
many of acid, and the fifth how many of water of com~
pofition. In fome cafes two proportions are given, and
it is expreffed in the third column under what ftate of
the fait thefe proportions exift.
LE II.
Decomposition by (Single Affinity.
Barytes.
Potafti.
Soda.
Lime.
Magnefia.
Ammonia.
Tannin.
Gallic Acid.
Oxalic Acid.
Tartaric Acid.
Vol. XVII. Part I,
Salt.
0
W H
H O
Ph
Hi
D
C/3
oi
M
Ph
to
to
Decomposition by Double Affinity.
Nitrate of Potafh.
Silver.
Muriate of Barytes.
• — Lime.
Soda.
Ammonia.
Carbonate of Barytes.
Lime.
Magnefia.
Potafh.
Soda.
Ammonia.
Acetate of Lead.
Subborate of Soda.
Rr
Previous
Kequifites.
Salt.
22. Carbonate of Magnesia.
23. Carbonate of Potash.
24. Subcarbonate of Potash.
25. Carbonate of Soda.
26. Carbonate of Ammonia.
27. Carbonate of Iron.
28. Carbonate of Zinc.
29. Acetate of Potash.
30. Acetate of Lead. Ph. Ed.
31. Subborate of Soda.
32. Sufertartrate of Potash.
33. Tartrate of Potash.
34. Tartrate of Potash and Soda.
35. Tartrate of Antimony and Potash, or Emetic
Tartar.
Solubility.
Infoluble.
6o°, 4.
212°, I.5
Deliquefcent.
Efflorefcent.
6o°, 2.
Infoluble.
Infoluble.
Deliquefcent.
6o°, 4.
6o®, 18.
2120, 6.
6o°, 60.
212°, 13.
_6o°, 4.
Deliquefcent.
6o°, 5.
Efflorefcent.
6o°, 15.
212°, 3.
Composition.
— A
Base.
45-
40.
Dry 64.
Cryft. 21.58
Dried 59.85
58.
I7*
33-
Tart. Pot.
54-
Ox. of Ant.
38-
Potafh 16.
Acid. Water.
34-
43-
3°-
14.42
40.05
26.
39-
67-
Tart. of
Soda.
46.
34-
21.
I7-
64.
o.
16.
44.
12.
29
Explana¬
tion.
313
Previous
Requifites.
In this firft part of the table of fecondary falls there
are five columns, in the firft of which are fet down the
names of moft of the fecondary falls employed in medi¬
cine, according to the moft approved chemical nomen¬
clature. The fecond column fhewsthe degree of attrac¬
tion which fubfifts between each fait and water, name¬
ly, how many parts of water at the temperatures of 6o°
and 212° of Fahrenheit are required todiffolve one part
of the fait, in the ftate in which it is ufually employed,
TAB
and whether the fait be deliquefcent or efflorefcent.
The three remaining columns point out, as far as has
been afeertained, the proportional quantities of the com¬
ponent parts of each fait, the third column (hewing how
many parts in the 100 confift of bafe $ the fourth how
many of acid, and the fifth how many of water of com~
pofition. In fome cafes two proportions are given, and
it is expreffed in the third column under what ftate of
the fait thefe proportions exift.
LE II.
Decomposition by (Single Affinity.
Barytes.
Potafti.
Soda.
Lime.
Magnefia.
Ammonia.
Tannin.
Gallic Acid.
Oxalic Acid.
Tartaric Acid.
Vol. XVII. Part I,
Salt.
0
W H
H O
Ph
Hi
D
C/3
oi
M
Ph
to
to
Decomposition by Double Affinity.
Nitrate of Potafh.
Silver.
Muriate of Barytes.
• — Lime.
Soda.
Ammonia.
Carbonate of Barytes.
Lime.
Magnefia.
Potafh.
Soda.
Ammonia.
Acetate of Lead.
Subborate of Soda.
Rr
Set display mode to:
![]() Universal Viewer |
Universal Viewer | ![]() Mirador |
Large image | Transcription
Mirador |
Large image | Transcription
Images and transcriptions on this page, including medium image downloads, may be used under the Creative Commons Attribution 4.0 International Licence unless otherwise stated. ![]()
| Permanent URL | https://digital.nls.uk/192353162 |
|---|
| Attribution and copyright: |
|
|---|
| Description | Ten editions of 'Encyclopaedia Britannica', issued from 1768-1903, in 231 volumes. Originally issued in 100 weekly parts (3 volumes) between 1768 and 1771 by publishers: Colin Macfarquhar and Andrew Bell (Edinburgh); editor: William Smellie: engraver: Andrew Bell. Expanded editions in the 19th century featured more volumes and contributions from leading experts in their fields. Managed and published in Edinburgh up to the 9th edition (25 volumes, from 1875-1889); the 10th edition (1902-1903) re-issued the 9th edition, with 11 supplementary volumes. |
|---|---|
| Additional NLS resources: |
|

